Quick Links: Study design | Patient demographics | Results | Proven safety | Proven effectiveness
Proven satisfaction | The power of Altis | Subscribe to emails | Important safety information

Patient after patient. Visit after visit. Three years of convincing data. Put your trust in Altis, the minimally invasive sling that can help your patients move beyond the daily limits of short-term solutions to experience long-term gains. The latest evidence shows at 36 months, Altis continues to perform similarly to full length retropubic and transobturator slings in treating stress urinary incontinence (SUI). Altis demonstrates similar performance in safety and efficacy1–delivering the proof you want and the solution your patients need.
Time after time
Altis 522 Post-Market Surveillance Study Design
![]()
Prospective, multi-center,
nonrandomized study
![]()
Women underwent
sling procedures
![]()
Physicians
![]()
Different sites (US & Canada)
Slings Tested
![]()
Single Incision Sling:
Altis
![]()
Comparator:
Full length retropubic
& transobturator slings
Timeframe Observed
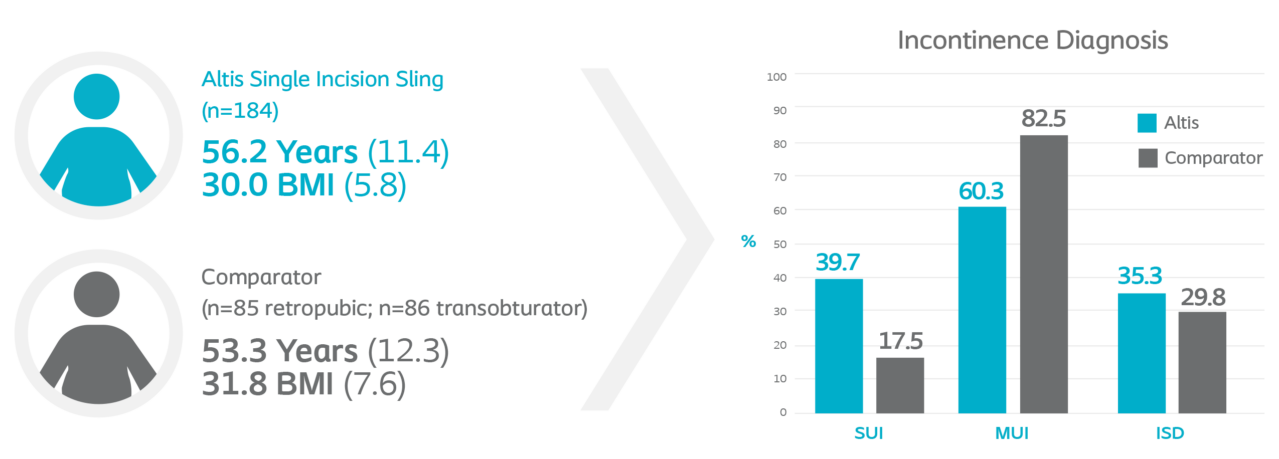
Altis 522 Study Patient Demographics2
Convincing Results.
Time After Time
Altis performs similarly to full length retropubic and transobturator slings in:
![]()
Safety
![]()
Effectiveness
![]()
Patient Satisfaction
Proven Safety.
Time After Time
The safety of your patients is your top priority and Altis consistently delivers. Designed to make procedures straight-forward, accurate and repeatable3,1, Altis demonstrated similar safety results to full length slings in the 36 month study.
Altis clinical performance data should provide the reassurance needed for your patients.
Altis vs Comparator
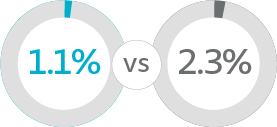
Device and/or procedure related serious adverse events (SAE)
Data for the treated population are presented as the number of subjects (%)
Compared to full length retropubic and transobturator slings, Altis:
- Had similarly low rates of device and/or procedure related serious adverse events
- Zero cases of mesh exposure, extrusion or erosion
Proven Effectiveness.
Time After Time
Altis provides the certainty you need with outcomes you expect. Data through 36 months reinforces reliability over time with Altis reporting similar efficacy as full length retropubic and transobturator slings. This evidence is more proof that Altis allows your patients to move beyond the limits of short-term solutions to experience long-term success.
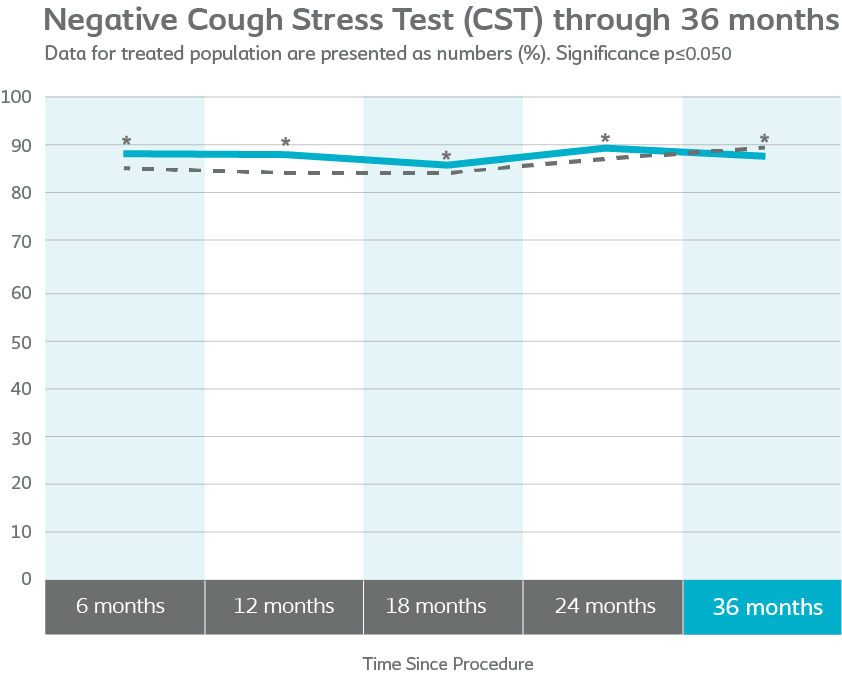

87.1% Altis®
CST demonstrated Altis was similar to retropubic and transobturator slings (p<0.001)
Altis performed similarly to the Comparator arm at every study visit and rates of negative CST remained above 80% for both groups.
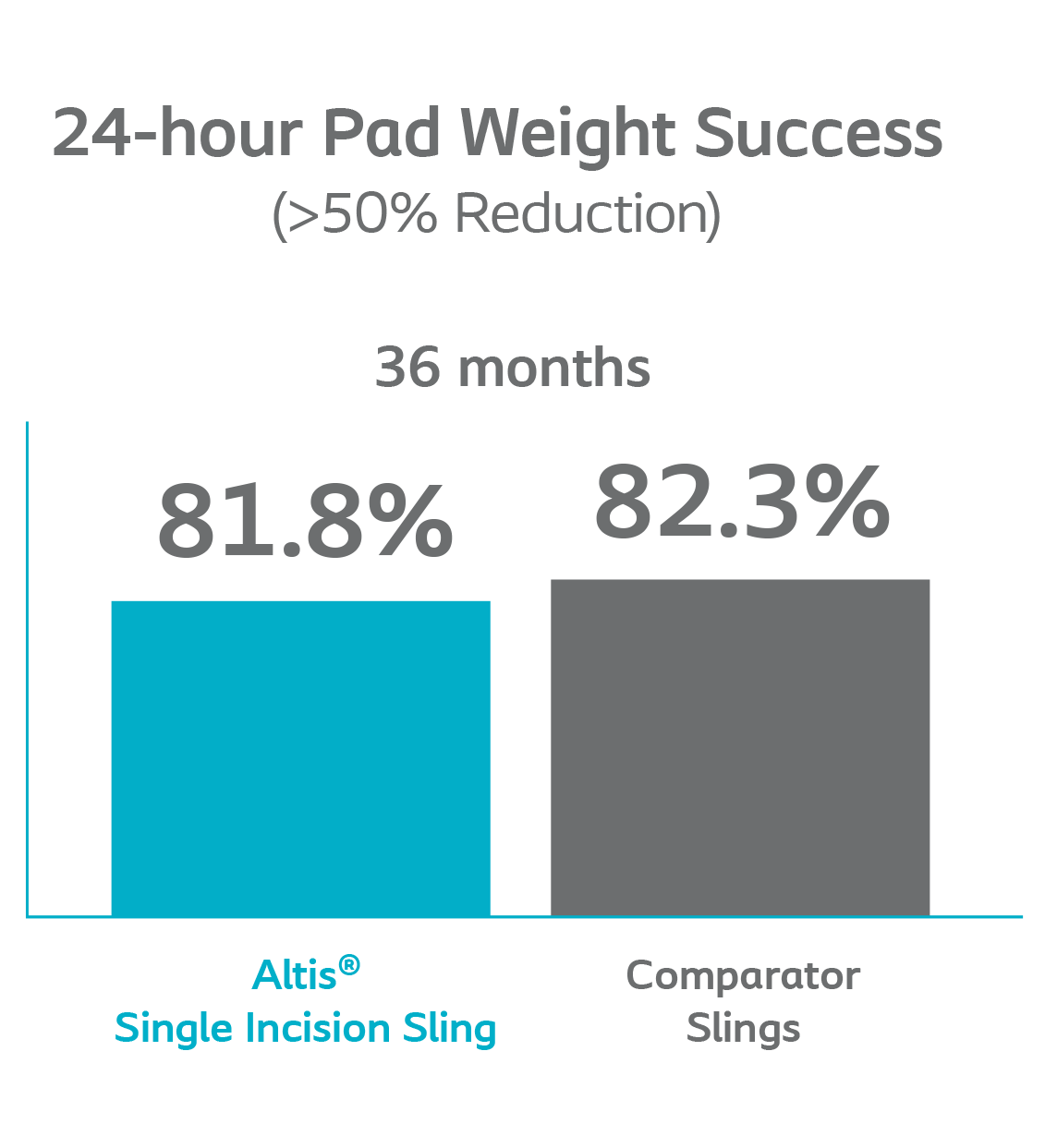
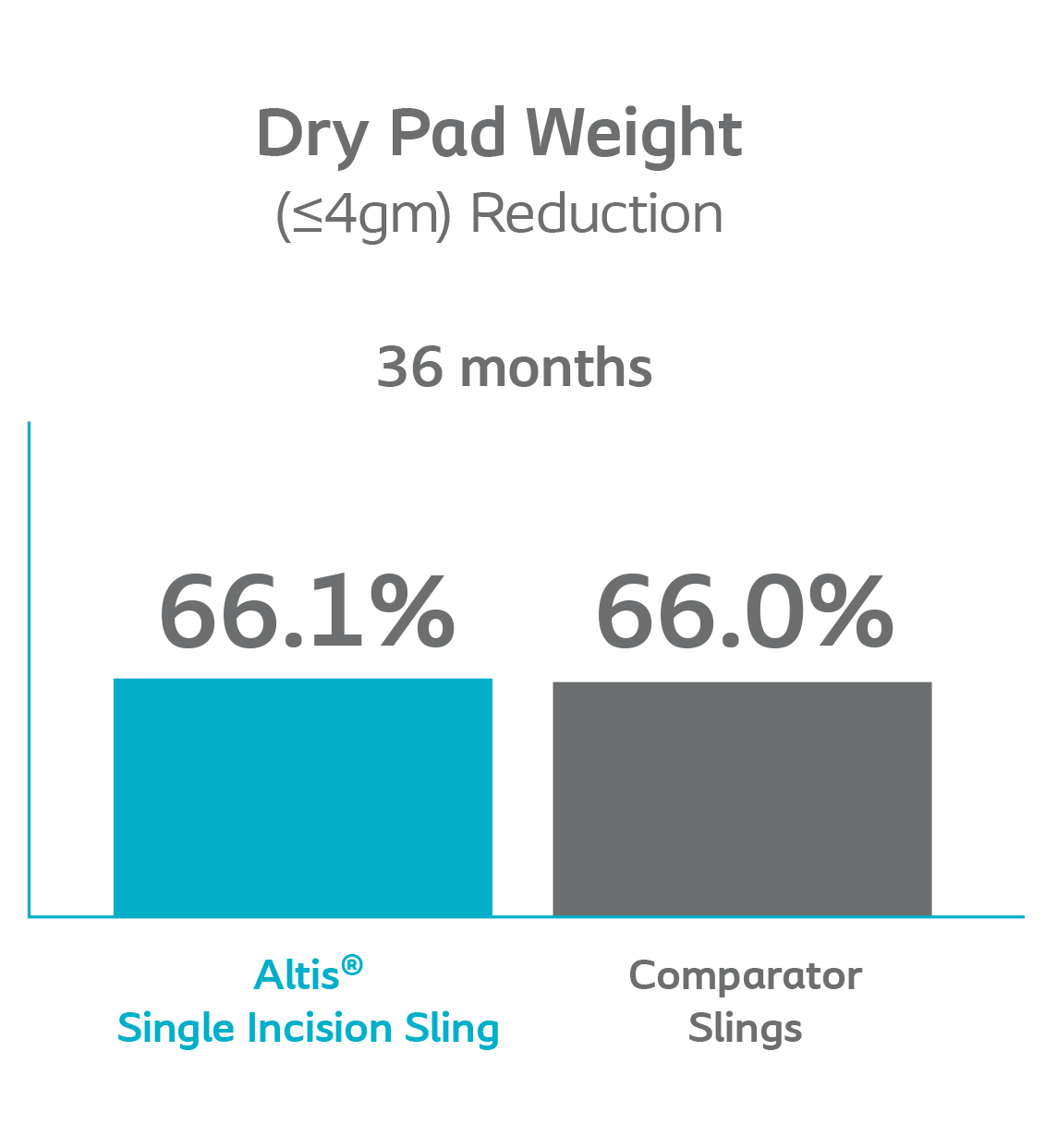
The rate of Altis patients with dry pad weight was similar to patients in the Comparator arm through 36 months.
Proven Satisfaction.
Time After Time
Altis is changing womens’ lives. Day in and day out. According to the latest research, Altis and full length sling patients had a high degree of satisfaction with their surgery. With an SUI treatment that’s more efficient and less invasive4, your patients need to know about the Altis alternative—and deserve a choice in their care.
| Patients had a high degree of satisfaction with their surgery5 |
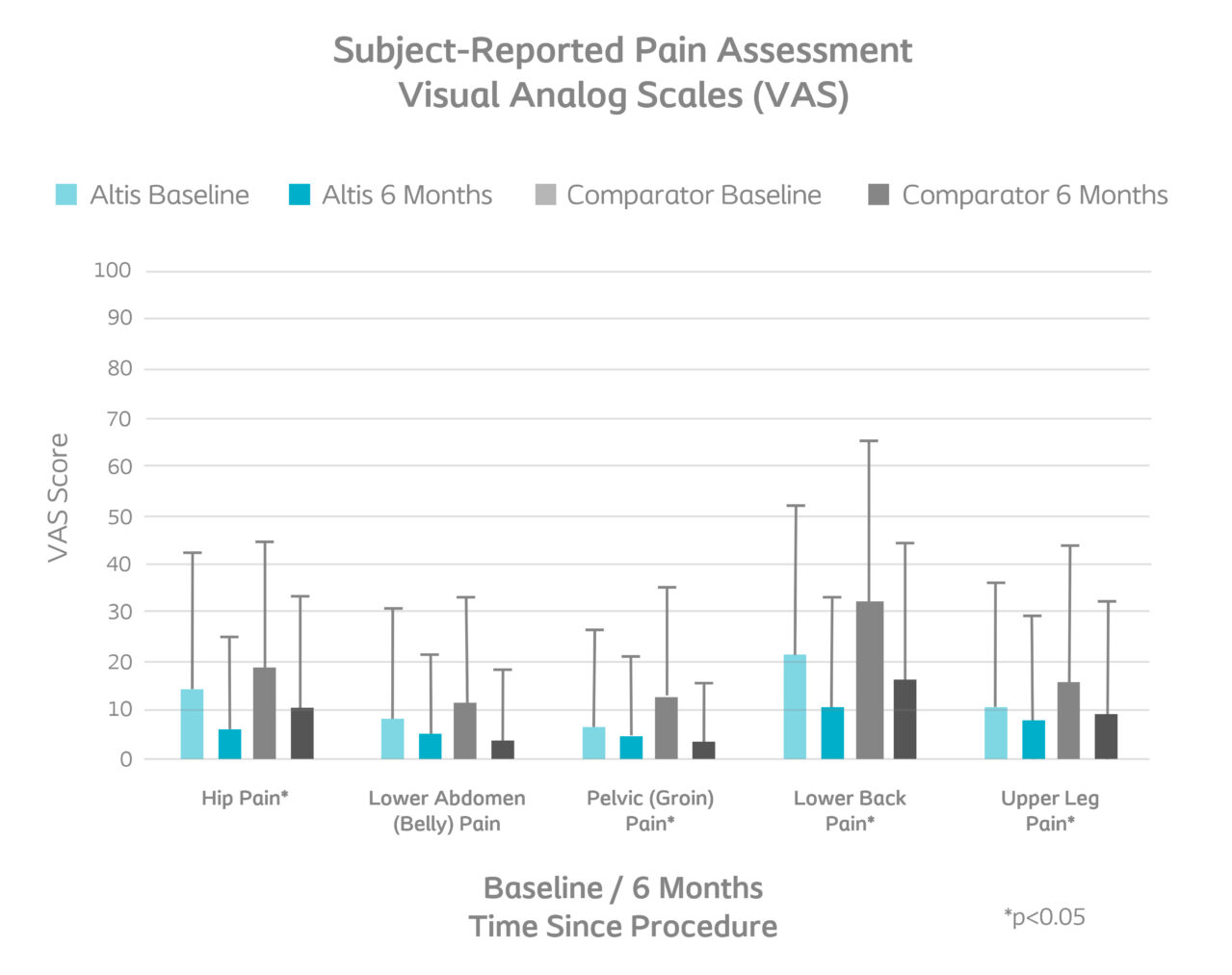
Pain decreased similarly5
|
| Improvement in quality of life and return to normal increased similarly5 |
| No difference in satisfaction in the amount of time to return to daily activities and work5 |
The Power of Altis.
Time After Time
LESS INVASIVE
One incision. Less tissue trauma. Speed up procedure time and deliver a better patient experience.3,4
STABLE SUPPORT
Patented, flexible, lightweight mesh is the thinnest available, allowing support of the urethra.3
REPRODUCIBLE INSERTION
Patented helical introducer makes the surgical procedure straightforward, accurate and reproducible.3
PRECISE TENSIONING
Place the device, then control the adjustable tensioning.3
RIGOROUSLY STUDIED
50+ publications. 3,000+ patients studied. The only sling
with a pre-market IDE and completed 522 study.

Request additional information from Coloplast Interventional Urology
Sign up to receive information and support. By opting into our emails, we can keep in touch with you on new updates.

