Saffron™ Fixation System
Designed for ease and reliability in prolapse repair
Quick Links: Product Detail | Product Resources | Ordering Information | Important Safety Information
Consistent, reliable deployment; ease of navigation; more choice and control
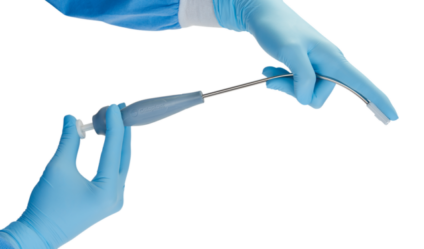
The Saffron Fixation System is indicated for attaching sutures to ligaments of the pelvic floor. It can be used with patients who are good surgical candidates for transvaginal surgical pelvic floor reconstruction. Saffron pairs consistent, reliable control and anchor deployment with ease of navigation, all while allowing surgeons to use their choice of suture and needles for POP repair. The design is intuitive, and easy to use, and surgeons report high satisfaction with the Saffron system.

Product Details
The two-piece system consists of one Saffron tool and biocompatible Saffron anchors (sold separately in boxes of 12). The Saffron tool is intended for multiple uses in a single surgical procedure. The anchors are intended to be permanently implanted and are for single-use only.
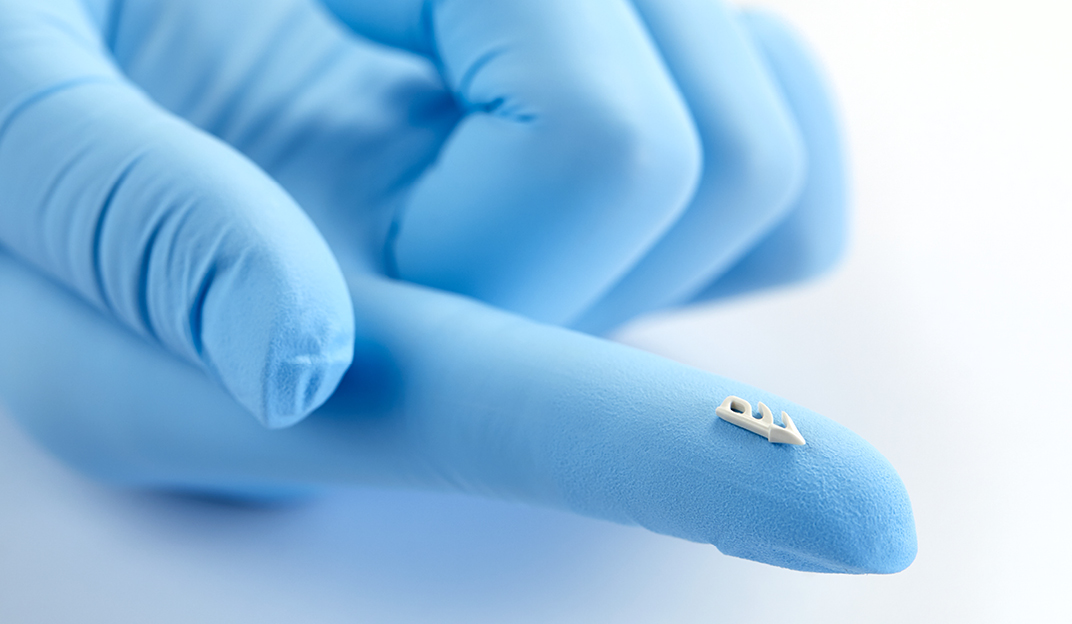
Smooth, secure anchor design with the smallest polymeric anchor volume available
Consistent, reliable deployment
- Intuitive design, easy to hold and deploy
- Better at maintaining distal tip placement than Capio,
Anchorsure and Digitex* - Design enables consistent anchor placement and precise anchor targeting
- Provides tactile confirmation that anchor has been deployed
- Fixation strength exceeds requirement for prolapse repair
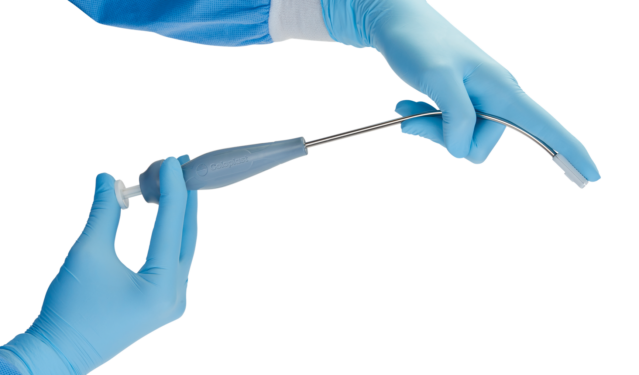
Ease of navigation
- Saffron’s unique curved design allows for easy navigation to the sacrospinous ligament
- Small distal tip for smooth entry into the dissection
- Design enables perpendicular placement on the ligament

More choice and control
- Use with your choice of commercially available sutures
- Accommodates most off-the-shelf sutures up to size 0 in any material or length (excluding barbed sutures)
- A wide array of needles can be used for the repair

Resources
Brochures
Saffron™ Fixation System Brochure Send a SMS Share
Saffron™ Fixation System Anchor Loading Guide Send a SMS Share
Saffron™ Fixation System Clinical Summary Send a SMS Share
Saffron™ Fixation System Infographic Send a SMS Share
Videos
Saffron™ Fixation System Surgical Video with Dr. Costas Apostolis, MD, FACOG, FPMRS Send a SMS Share
Ordering information
For ordering information, please call 800-258-3476.
Saffron™ Fixation System
| Saffron™ Fixation System | Purchase UOM | Qty per UOM | Item |
| Saffron Fixation Tool | EA | 1 | 520340 |
| Saffron Anchor | Box | 12 | 520350 |
