INTIBIA™
Implantable Tibial Nerve Stimulator
A patient with the Coloplast Intibia Tibial Nerve Stimulator, given that the lead is connected to the IPG, may be safely scanned under the following conditions. Failure to follow these conditions may result in injury to the patient.
| MR Conditional | |
| Device Name | Coloplast Intibia Tibial Nerve Stimulator |
| MRI Systems | 1.5 T or 3 T |
| MR Scanner Type | Cylindrical |
| B0 Field Orientation | Horizontal |
| Maximum Spatial Field Gradient | 20 T/m (2000 gauss/cm) |
| Maximum Gradient Slew Rate | 200 T/ms |
| RF Excitation | Circularly Polarized (CP) |
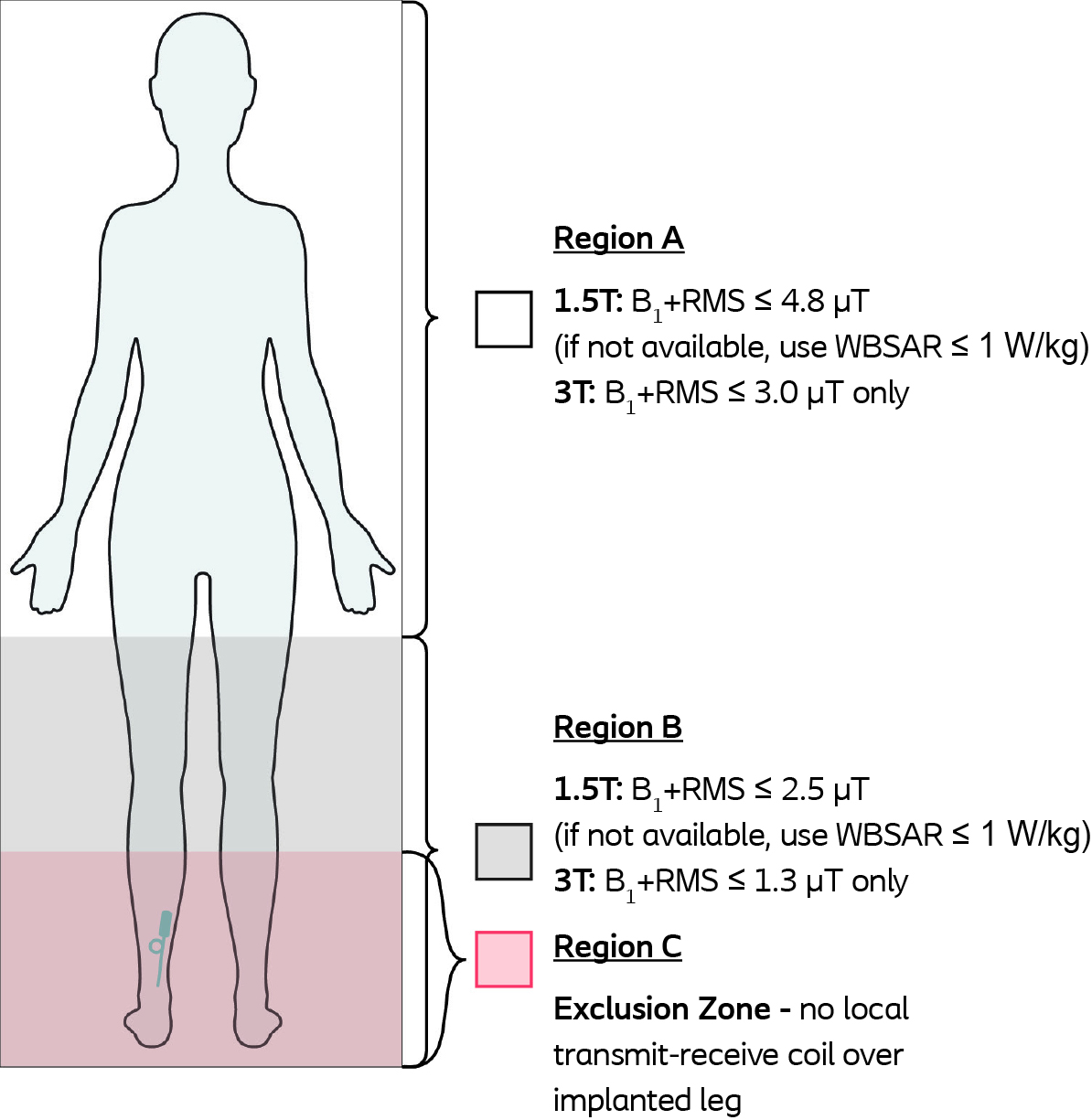
| RF Transmit Coil Type | Integrated Whole Body Transmit Coil: Any anatomical region is acceptable. See RF conditions below. These conditions also apply when using local receive-only coils. Local Transmit-Receive Coils: Regions A and B only; do not use local transmit-receive coil on implanted leg in Region C. |
| Operating Mode | Normal Operating Mode |
| Maximum Head SAR | 3.2 W/kg |
| RF Conditions | See diagram |
| Scan Duration | 60 minutes of continuous RF (a sequence or back-to-back series/scan without breaks). |
| MR Image Artifact | The presence of this implant may produce an image artifact of 38 mm |
| If information about a specific parameter is not included, there are no conditions associated with that parameter. | |
Ultrasound: The INTIBIA ITNS should not be exposed to therapeutic levels of ultrasound energy, as the implantable part of the active implantable medical device can inadvertently concentrate the ultrasound field and cause harm.
Radiation: Electronic components can be damaged by therapeutic ionizing radiation, and any damage may not be immediately detectable.
Environmental hazards: The INTIBIA ITNS complies with ISO 14708-3:2017 Clause 27 and IEC 60601-1-2 Table 11 for protection from electromagnetic interference (EMI). A strong magnet aligned in proximity to an INTIBIA ITNS will stop therapy. Timed applications of a strong magnet can program an INTIBIA ITNS when its programmer is unavailable. Patients should be advised to seek medical guidance if any unusual sensation occurs at the implant site during exposure to an EMI source.
Other active implantable medical devices: The INTIBIA ITNS has not been tested for interference with other electronic implants such as pacemakers, defibrillators, and neurostimulators. If the INTIBIA ITNS is to be used with other electronic implants, an interaction study must be performed by the physician.
*Caution: Investigational device. Limited by Federal (or United States) law to investigational use.
*This medical device is currently not for sale in any country.